LONG-TERM FEEDING WITH BIOACTIVE TRIPEPTIDES IN AGED HYPERTENSIVE AND NORMOTENSIVE RATS: SPECIAL FOCUS ON BLOOD PRESSURE
AND BRADYKININ-INDUCED VASCULAR REACTIVITY
2Metropolia University of Applied Sciences, Helsinki, Finland.
INTRODUCTION
High blood pressure causes cardiovascular diseases and increases the risk of premature death (WHO: Prevention of Cardiovascular Diseases 2007). Bioactive compounds present in many foods e.g. in milk, egg, berries and fish, might offer a way to prevent the development of high blood pressure or even reduce already slightly elevated blood pressure (1-3). Indeed, when milk is fermented with certain bacteria, its proteins degrade into bioactive peptides like isoleucine-proline-proline (Ile-Pro-Pro), leucine-proline-proline (Leu-Pro-Pro) and valine-proline-proline (Val-Pro-Pro) (1). These peptides have been demonstrated to decrease elevated blood pressure in both humans (4-7) and in experimental models (8). Irrespective of the apparent blood pressure lowering effect in sub-chronic clinical trials (4-7) with slightly hypertensive but not with normotensive subjects and strong antihypertensive evidence from animal experiments (8-11), the European authorities have not approved the status of functional food for the peptide containing products (e.g. Calpis, Evolus). The main mechanism of action is believed to be inhibition of angiotensin-converting enzyme 1 (ACE-1) (12). However, these compounds seem to have also other modes of actions, possibly inhibiting other enzymes (12, 13), or alternatively regulating receptor activation (14), which may at least partly contribute to their overall beneficial effects on blood pressure and vasculature. Fermented milk contains also a variety of other bioactive ACE-1 inhibitory peptides (15, 16).
Under normal conditions, bradykinin acts via two different receptors, bradykinin receptor type 1 (BR1) and type 2 (BR2); it induces vasodilatation via nitric oxide (NO), prostacyclin and endothelium-derived hyperpolarizing factor (EDHF) (17). The expressions of BR1 and BR2 receptors as well as bradykinin’s ability to activate these receptors decrease with ageing and in inflammation (14, 18-23) i.e. with ageing, bradykinin induced vasodilatation declines or it can even be converted into vasoconstriction (21). We have previously shown that a bioactive tripeptide, Ile-Pro-Pro, can potentiate bradykinin-induced vasodilatation, especially in blood vessels from aged animals; this phenomenon is related to the Ang(1-7)-Mas-receptor axis (14, 24).
In the present study, we investigated the effects of long term tripeptide feeding on young and aged spontaneously hypertensive rats (SHR) and age-matched normotensive rats (WKY), focusing especially on blood pressure development and bradykinin-induced vascular functions.
MATERIAL AND METHODS
Experimental protocol
Four and eighteen weeks old spontaneously hypertensive rats (SHR, 8 animals in each group) and age-matched Wistar-Kyoto rats (WKY, 6 animals in each group) were fed for 6 weeks ad libitum with a tripeptide-containing drink. Rats were housed 2 – 4 animals in a cage. The study protocol was approved by the National Animal Experimentation Committee of Finland according to EC Directive 86/609/ECC and the Finnish Experimental Animal Act 62/2006s.
Experimental drink
The tripeptide-containing (0.1 mg/ml) drink was prepared by mixing tripeptide enriched powder (Valio Ltd, Helsinki, Finland) into tap water. The powder contained Ile-Pro-Pro 7200 mg/kg, Val-Pro-Pro 5800 mg/kg and Leu-Pro-Pro 3200 mg/kg (analyzed using LC-MS method by Valio Ltd). Animals drinking tap water were used as the control. The consumption of the drinking fluid was measured daily and feed consumption (Teklad Global 16% Protein Rodent Diet, Harlan Laboratories, Cambridgeshire, United Kingdom) weekly.
Blood pressure measurement
Systolic and diastolic blood pressure (SBP, DBP) were measured weekly by the tail-cuff method (CODA non-invasive blood pressure-equipment, Kent Scientific Corporation, Connecticut, USA) by the same researcher (AS) at the same time of the day (between 8 – 12 am.). Animals were warmed in a warm cabinet at 32°C for 20 min before measurements to make the pulsation of tail artery more readily detectable. Blood pressure was recorded at least 8 times in 3 different sets. The mean of all measurements (at least 3) accepted by the CODA program were calculated, and these values presented SBP and DBP level of the week.
Vascular reactivity measurement
After anesthesia (isoflurane 5%, Vetflurane®, Virbac, Carros, France), blood was collected by heart puncture, and the animals were decapitated. Plasma was isolated from whole blood in heparin-containing tubes (Venosafe®, Terumo, Leuven, Belgium) by centrifugation (1500 g, 10 min, 4°C) and stored at –80°C. The descending aorta was isolated, cleaned from adherent tissues and stored at –80°C prior to analysis. Superior mesenteric arteries were isolated and placed in ice-cold pre-oxygenated (95% O2/5% CO2, Aga Oy, Riihimaki, Finland) Krebs buffer (NaCl 119 mM, NaHCO3 25 mM, glucose 11.1 mM, KCl 4.7 mM, CaCl2 1.6 mM, KH2PO4 1.2 mM and MgSO4 1.2 mM). Adherent adipose and connective tissue was carefully removed under a light microscope (Leica E24 HD, Wetzlar, Germany), and the arteries were cut into 3 mm rings and hooked using stainless steel hooks. Hooked artery rings were placed in constantly oxygenized Krebs buffer (37°C) in tissue chambers and vascular reactivity was measured using with a computer-linked myograph (EMKA Technologies, Paris, France).
The protocol for vascular reactivity studies was as follows: after one hour equilibration with 1.5 g baseline tone, the arteries were exposed to 60 mM potassium chloride (KCl) so that they would contract to obtain a measure of non-receptor mediated vasoconstriction. Functionality and intactness of the endothelium was measured using cumulative dosing with acetylcholine (ACh 1 nM – 10 µM, Sigma-Aldrich, Saint Louis, USA) after phenylephrine (PE, 1 µM, Sigma-Aldrich) constriction. The role of bradykinin-induced vascular reactivity (bradykinin acetate salt, BK 1 nM-10 µM, Bachem, Weil am Rhein, Germany) was measured with or without inhibitors, agonists or antagonists against different enzymes and receptors after PE constriction. Diclofenac (3 µM; Sigma-Aldrich) was used as a nonselective cyclooxygenase (COX) inhibitor, A-779 (0.1 µM, Bachem) as a Mas-receptor antagonist and noscapine (1 µM, Sigma-Aldrich) as a non-subtype selective bradykinin receptor (BR1 and BR2) antagonist. Vascular rings were washed at least three times and equilibrated for 20 – 30 min between different experimental sets. All the concentrations mentioned here are the final concentrations in the chambers.
qPCR experiments
The levels of mRNA of COX-1, COX-2, ACE-1, BR1, BR2 and Mas-receptor were measured from the aortas of the rats. β-actin, glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and lactate dehydrogenase A (LDHA) were used as control genes. The mRNA isolation protocol was as follows: about one cm section of descending aorta was cut into pieces with a razor blade and ground into powder in liquid nitrogen using a mortar and pestle. The mRNA isolation was performed with commercially available Trizol reagent (InVitrogen, Thermo Fisher Scientific, MA, USA) according to the manufacturer’s instructions. Briefly, 0.5 ml Trizol reagent was added and the sample was homogenized (5500 rpm, 3 × 20 s, Precellys 24, Bertin Technologies, Montigny le Bretonneux, France). After homogenization, mRNA was extracted using chloroform and washed with isopropanol and ethanol. At the end of the protocol, mRNA was diluted into nucleic acid free water and possible remnants of DNA were removed using DNAse enzyme (Machery-Nagel, Duren, Germany). The total amount of RNA was measured in a NanoDrop spectrophotometer (Thermo Fisher Scientific). One µg of RNA was converted to cDNA with a commercially available kit (iScript, BioRad, CA, USA) following the instructions provided by the manufacturer.
All samples were diluted 1:5 using nuclei acid free water. Two to four µl of diluted samples, 1 µl of each primer (forward (F) and reverse (R)), 10 µl of MasterMix (LightCycler 480 SYBR Green I Master, Roche, Mannheim, Germany) and 4 – 6 µl of nuclei acid free water were used in the final qPCR experiments (altogether 20 µM per reaction). qPCR analyses were performed with a LightCycler 480 Instrument II (Roche). Parameters were as follows: Initial 10 min in 95°C, followed by 40 cycles of denaturation (15 s, 95°C), annealing (30 s, 60°C, except 58°C for Mas-receptor) and elongation (30 s, 72°C). Melt curve analysis was performed at the end of experiment (5 s, 95°C, 1 min 60°C followed by temperature increases at 0.11°C/s until 95°C). The efficiency of the primers was tested before actual qPCR analysis using dilution serial from the pooled samples. The primers (‘5→3’) used for the analysis were:
ACE-1 F: AGTGGGTGCTGCTCTTCCTA and
R: ATGGGACACTCCTCTGTTGG,
COX1 F: TCCGTGAAGATGCGCTACC and
R: AACACCTCCTGGGCCACAG (adapted from Cleveau et al. (25)),
COX2 F: CATGATCTACCCTCCCCACG and
R: CAGACCAAAGACTTCCTGCCC (adapted from Cleveau et al. (25)),
B1 F: AGGGTTCGTCATCACTAT and
R:AGGTAGATTTCCGCTATG, B2 F: TGAGGAACAACGAGATGAAGAAG and
R: GGAAACCAACACAGCACAAAGAC (adapted from Sang et al. (26)),
PGIS F: GACGTTTTCCGCACCTTCC and
R: ACTGACAAGGAGCCTCGAGC (adapted from Cleveau et al. (25),
Mas F: TGTGGGTGGCTTTCGATTT and
R: ATTAGACCCCCATGCATGTAGAA (adapted from Xue et al. (27)),
β-actin F: AGATCAAGATCATTGCTCCTCCT and R: AAAACGCAGCTCAGTAACAGT,
GAPDH F: GCTGCCTTCTCTTGTGACAA and R: ATCTCGCTCCTGGAAGATGG and
LDHA F: CATCGTGCATAAGCGGTCC and R: GCAAGCTCATCAGCCAAGTC.
Primers were designed using Primer Blast software (NCBI) if not mentioned otherwise. Results were calculated using the method of Vandersompele et al. (28). qPCR measurements were performed only for young and old SHR and WKY consuming water, thus tripeptide-consuming groups were omitted for technical and economic reasons.
ACE-1 activity assay
ACE-1 activity was measured using the method of Schwager et al. (29) with minor modifications. Briefly, isolated protein samples from aorta (200 µg/ml) were used. Activity was measured using the substrate, hippuryl-L-histidyl-L-leucine (HHL). ACE-1 cleaves the substrate to expose a free N-terminus, which can be fluorogenically labeled with o-phthaldialdehyde (OPA). In the first reaction HLL (6 mM, Sigma-Aldrich) was incubated for 10 min in 37°C with sample or standard (HL, Sigma-Aldrich) followed by the second reaction with OPA (1 mg per reaction in ethanol, Sigma-Aldrich) in 10 min in dark at room temperature. The reaction was stopped using HCl (3 M, Sigma-Aldrich) and fluorescence (ex 355nm/em 535 nm) was measured using Wallac Victor 1420 multilabel counter (Perkin Elmer, Turku, Finland). Captopril (10 µM, Sigma-Aldrich) was used as a control using pooled samples.
Western blot
Segments of kidneys were homogenized in Elisa buffer (NaCl 136 mM, Na2HPO4 8 mM, KCl 2.7 mM, KH2PO4 4.46 mM, Tween 0.001%, pH 7.4) containing protease inhibitor cocktail set X (Calbiochem, Merck Millipore, Darmstadt, Germany). Supernatants containing proteins were collected and the amount of total proteins was measured with a commercial kit (Pierce™ BCA Protein Assay Kit, Thermo Fisher Scientific, Waltham, USA). Samples were diluted into Laemmlis buffer (Bio-Rad). Samples containing 20 µg protein were fractioned in 7.5% poly-acrylamide gel in SDS-Page and transferred onto nitrocellulose membrane (Bio-Rad). Membranes were blocked using commercially available buffer (Odyssey Blocking Buffer, LI-COR, Lincoln, NE, USA) incubated with primary antibodies (1:50 ACE-1, 1:2000 BR2, 1:250 COX-1 (Thermo Fischer), BR1 1:1000 (Santa Cruz Biotechnology, Heidelberg, Germany ) and 1:3000 β-actin (Cell Signaling Technology, Danvers, MA, USA)) overnight at 4°C followed by 1 hour fluorescein-labeled secondary antibody (IRDye680LT goat anti-mouse, IRDye800CW goat anti-rabbit, LI-COR) incubation at room temperature. Detection of the protein bands was performed using Odyssey CLx InfraRed Imaging system (LI-COR). β-actin was used as a loading control. We also tried to measure COX-2 protein levels using three different antibodies. However, we were not able to get reliable results from measurements, not even when positive control was used.
Biochemical measurements
The levels of ACE-1 activity and cyclic guanosine-3´-5´- monophosphate (cGMP, cGMP Elisa kit, Cayman Chemical, MI, USA) were measured from isolated proteins of descending aorta. Total nitric oxide (NOx, Nitrate/Nitrite colorimetric assay kit, Cayman Chemical) and cytokines (Q-Plex array rat, Quansys Bioscience, Utah, USA) were measured from plasma. Before the NOx assay, plasma samples were filtered through 30 kDa filters (30 k MWCO Spin-X, Corning).
Total proteins were isolated as followed: one cm piece of aorta was homogenized (5000 rpm, 3X20 s, Precellys 24, Bertin Technologies) with 100 mM Tris-0.1% Triton X buffer using pearl beads. After homogenization, the samples were incubated for 15 min in ice and centrifuged at 12,000g for 15 min in 4°C. Supernatants were collected and stored at –80°C. Total protein concentrations were measured with a commercial kit (Pierce™ BCA Protein Assay Kit, Thermo Fisher Scientific). All samples were diluted into 100 mM Tris-120 mM NaCl buffer to protein concentration 200 µg/ml.
Statistical analyses
Statistical analyses were performed using Student t-test (two tailed) or one-way ANOVA followed by Tukey’s or Dunnett’s post-hoc test depending on the number of groups being compared. The tests were done using GraphPad Prism software (version 5). P-values less than 0.05 were considered as a statistically significant difference. All data are presented as mean ± S.E.M. except for the qPCR result which is a geometrical mean with maximum and minimum.
RESULTS
Weights, drink and feed consumptions
Weight gain, organ weights at the end of the experiment, fluid, peptide and feed consumption throughout the experiment are collated in Table 1. The weights of heart of the hypertensive animals were heavier than those of normotensive animals (P < 0.05). Young hypertensive animals had heavier kidneys than their young normotensive animals and old hypertensive animals (P < 0.05). Old animals drank more than young animals in each group (P < 0.05). However, if the total amount of daily peptide intake was divided by the body weight of the rat, the intake was higher in the young groups (P < 0.05). Rats also consumed more peptide-containing drink than water (P < 0.05). It seems that rats prefer the taste of the peptide- or protein containing drinks more than plain water and drink therefore more than their physiological need. This has been the fact also in earlier studies with similar experimental setup (10).
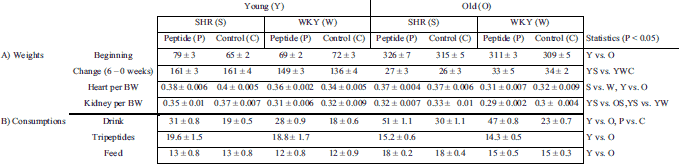
Blood pressure
The mean arterial pressure (MAP) of the groups is presented in Fig. 1. In old SHR, MAP was reduced at the end of the experiment in the peptide group (P < 0.05). In young animals, there were no differences between MAP values at the end of the six-week study period.
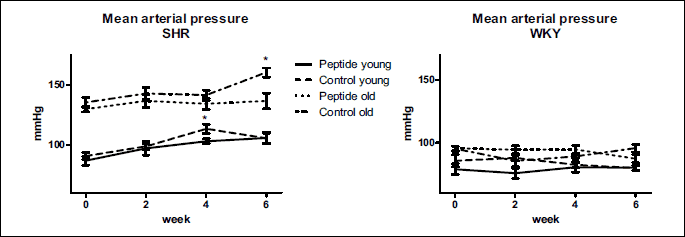
In young SHR, there was no difference between systolic or diastolic blood pressure (SBP and DBP) levels at either the beginning or the end of the experiment (Table 2). Blood pressure was slightly elevated in both groups. Blood pressure levels of young WKY remained stable throughout the experiment, and there was no difference between the groups. In old SHR peptide group, BP became only slightly elevated during the experiment (change between the beginning and the end: SBP 5 ± 5 and DBP 7 ± 8 mmHg). In the old hypertensive control group, blood pressure elevated more than in the peptide group during experiment (change between the beginning and the end: SBP 22 ± 6 and DBP 26 ± 8 mmHg, P < 0.05 in both). In the old WKY peptide drinking group, BP levels decreased during experiment (SBP –11 ± 3 and DBP –9 ± 3 mmHg, P < 0.05 in SBP). This blood pressure reduction was not evident in the control group (DBP 1 ± 4 and SBP –1 ± 4 mmHg).
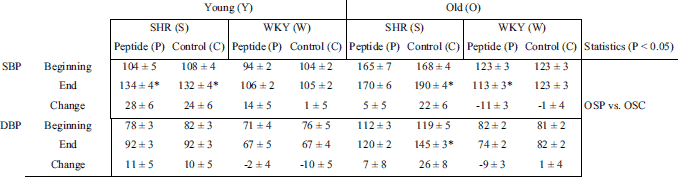
Vascular reactivity
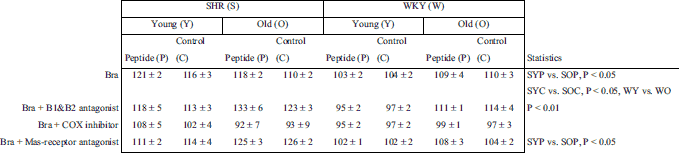
The endothelium-dependent vasodilatation was impaired in the aged SHR, apparently because of endothelial dysfunction, a phenomenon not detected in the old WKY (Fig. 2). PE- (receptor mediated) and KCl-induced (non-receptor mediated) vasoconstriction was lower in the groups of aged SHR, indicating that there was some dysfunction also present in smooth muscle cells (Fig. 2). There were no differences in endothelial or smooth muscle function between the normotensive groups either in terms of vasodilatation or vasoconstriction. Tripeptide consumption did not influence either endothelium or smooth muscle function.
In hypertensive animals, bradykinin induced vasoconstriction both in young and old animals (Table 4). In the normotensive groups, bradykinin caused also a minor vasoconstriction although less so than in the hypertensive rats (Table 4).
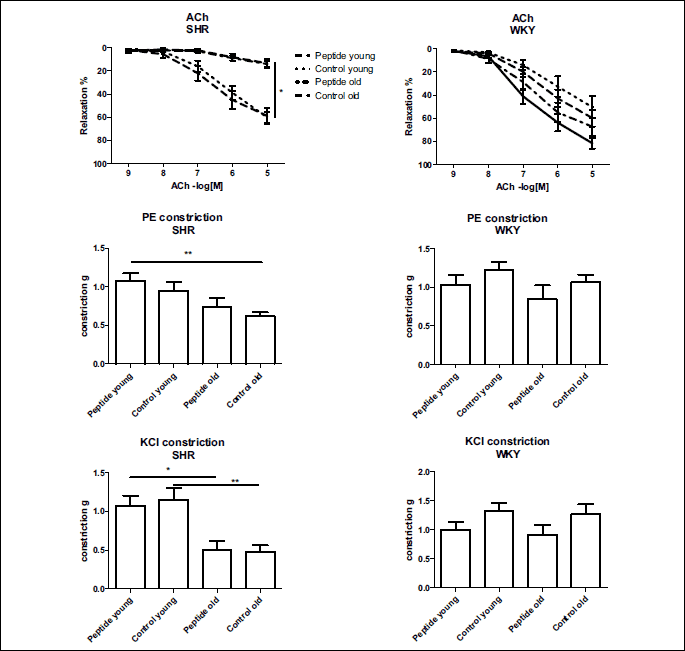
Mechanisms of bradykinin-induced vasoconstriction were evaluated using bradykinin receptor antagonists, a nonselective COX inhibitor or a Mas-receptor antagonist before bradykinin cumulative dosing. After COX inhibition, bradykinin-induced vasoconstriction was abolished in all of the groups (P < 0.05 compared to control) (Fig. 3 and 4). In the young normotensive groups, vasoconstriction was abolished after pretreatment with the bradykinin receptor antagonist (P < 0.05) (Fig. 4A and 4C). However, this was not seen in hypertensive or old groups. Pretreatment with the Mas-receptor antagonist had no effect on bradykinin’s vasoactivity.
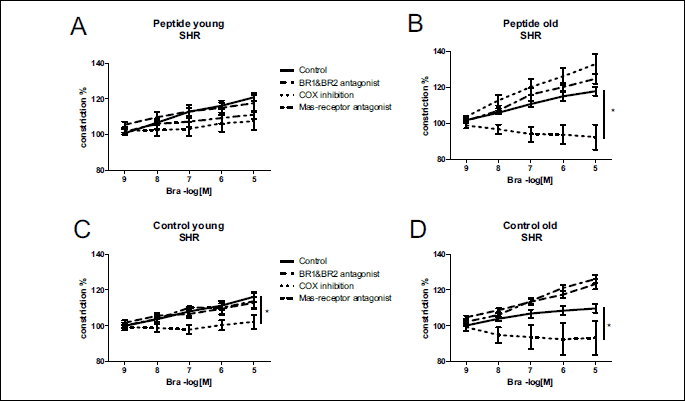
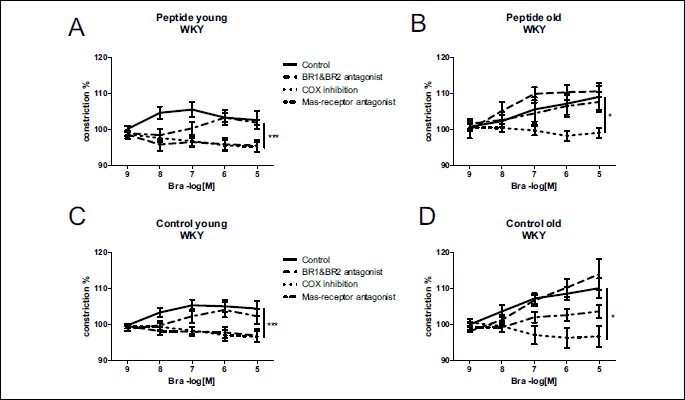
In the young SHR control group, the bradykinin-induced vasoconstriction after blockade of both bradykinin receptors was lower than in the old control group (P < 0.05) (Table 4). A similar phenomenon was seen after the Mas-receptor antagonist when comparing between the peptide consuming groups (P < 0.05) (Table 4). In the normotensive groups, bradykinin-induced vasoconstriction was smaller in young animals compared to their older counterparts (P < 0.05) (Table 4). There was no difference between groups after COX inhibition, i.e. vasoconstriction was abolished equally in all groups.
ACE-1 activity
ACE-1 activity in aorta was higher in groups of old animals both in the normotensive and the hypertensive groups (P < 0.001) (Fig. 5). Captopril, a potent ACE-1 inhibitor, blocked in vitro ACE-1 activity almost completely, whereas peptide feeding exerted no effect on ACE-1 activity.
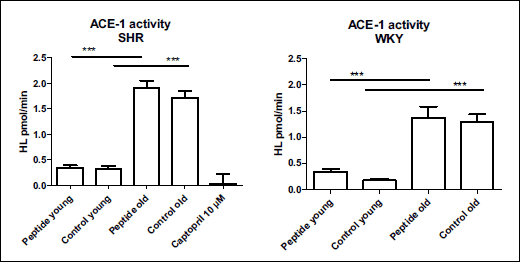 |
Fig. 5. Angiotensin-converting enzyme 1 (ACE-1) activity in aorta in different groups. Captopril was used as a negative control using pooled samples from all groups. Mean ± S.E.M., n = 6 – 8/group, ***P < 0.001. |
qPCR
Relative mRNA expression of cyclooxygenase (COX) enzymes seemed to be elevated in old hypertensive animals compared to their normotensive counterparts (P ns, Fig. 6A and 6B). In particular, COX2 expression was 5 times higher in old SHRs compared to normotensive WKY groups (P ns.) (Fig. 6B). Prostacyclin synthase (PGIS) expression was higher in old SHR than in old WKY (P < 0.05) (Fig. 6G). Unexpectedly, there were no differences in the relative ACE-1 expressions between the study groups (Fig. 6D) nor were any differences detected between BR1 and BR2 expression (Fig. 6E or 6F). Mas-receptor expression was highest in the young SHR (P < 0.05), a phenomenon not seen in old SHR (Fig. 6C).
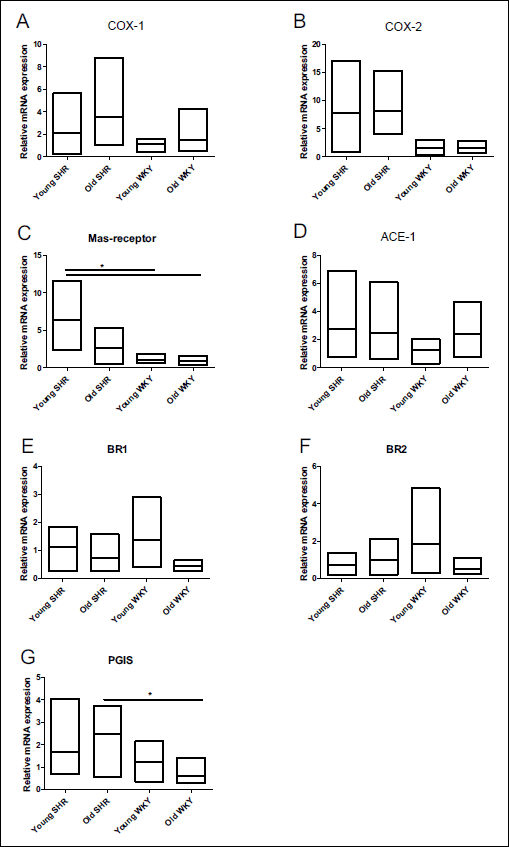 |
Fig. 6. COX-1 (A), COX-2 (B), Mas-receptor (C), ACE-1 (D), BR1 (E), BR2 (F) and PGIS (G) relative mRNA expression in aorta between groups. Data is presented as geometrical mean with maximum and minimum, n = 4 – 6/group, *P < 0.05. |
Western blot
Protein levels of ACE-1, COX-1, BR1 and BR2 were measured from kidney extract of young and old SHR and WKY (Fig. 7). β-actin was used as a loading control and the results are generated by dividing intensity of the band of measured protein by b-actin intensity from the same sample. ACE-1 protein levels were higher in old animals (Fig. 7A, P < 0.001). COX-1 and BR1 levels were slightly higher in hypertensive animals (Fig. 7B and FC). BR2 receptor protein level was similar in all investigated groups (Fig. 7D).
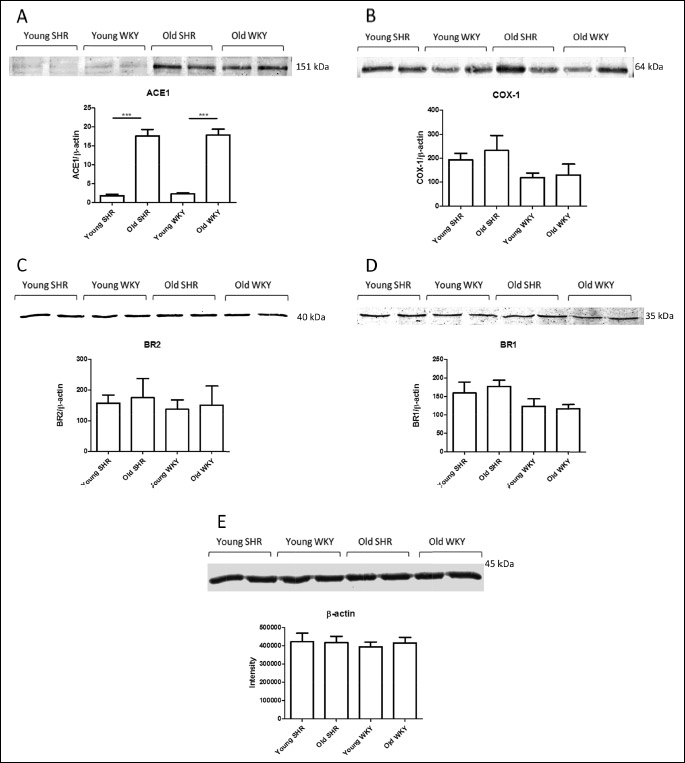
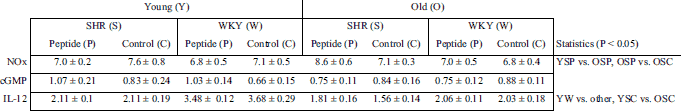
Biochemical measurements
The results from the NOx, cGMP and IL-12 measurements are presented in Table 3. Peptide consumption increased the plasma total nitric oxide (NOx) concentration in old SHR compared to the controls (P < 0.05). There were no differences in cGMP or TXB2 (data not show) levels between the groups. IL-12 levels were higher in young normotensive animals compared to all other groups (P < 0.001). In SHR, IL-12 levels were lower in old controls compared to their younger counterparts (P < 0.05). Peptide feeding did not influence any of the groups.
In the 9-Plex cytokine Elisa assay kit, only interleukine-12 (IL-12) was over the assay’s limit of quantification (74.20 pg/ml). All the other measured cytokines, IL-1a (22.23 pg/ml), IL-1β (27.27 pg/ml), IL-2 (111.28 pg/ml), IL-4 (9.15 pg/ml), IL-6 (231.32 pg/ml), IL-10 (10.28 pg/ml), IFNγ (2152.49 pg/ml) and TNF-α (41.88 pg/ml) were less than the lower limit of quantification (program Q-View, version 3.07, Quansys Bioscience). Lower limits of quantifications are indicated in brackets.
DISCUSSION
The present study had two major aims; firstly to compare blood pressure and vascular effects of long term feeding with bioactive ACE-inhibitory tripeptides in young and old normotensive and spontaneously hypertensive rats. Secondly, the mechanisms underpinning these effects at the vascular level were evaluated focusing on age, endothelial function and bradykinin.
Already at a young age, SRH showed clearly higher blood pressure than the normotensive WKY. The old SHR developed severe hypertension while the old normotensive animal had only slightly higher blood pressure than their respective young rats. Long-term tripeptide feeding inhibited the increase in blood pressure in the old SHR in comparison with the controls as we have shown in previous studies (8). The tripeptides lowered the blood pressure of young hypertensive animals after 4 – 5 weeks’ feeding. However, the difference was not statistically significant after 6 weeks’ feeding when compared with their water drinking littermates. For that reason, the blood pressure lowering effect in the young hypertensive animals remained unclear. Previously, tripeptide feeding lowered blood pressure also in younger animals (8-10). However, those animals were somewhat older than the young rats in the present study and furthermore both the tripeptide concentrations and the duration of the feeding were different from this study. In the old normotensive animals but not in the young rats, tripeptide feeding lowered blood pressure levels compared to the situation at the beginning of the study. This is the first time in our studies that tripeptide feeding has been demonstrated to reduce the blood pressure of normotensive animals. However, in previous studies, normotensive animals were younger than in our present study.
We and others have shown earlier that bradykinin-induced vasorelaxation is age-dependent (14, 21). This age-dependency of bradykinin’s cardiovascular actions is believed to be attributable to changes in the density of the bradykinin receptors. The numbers of the receptors decline during ageing and this lessens bradykinin’s protective effect on the heart and vasculature (19, 20, 22, 23, 26, 30-31). However, in this study we were not able to see difference in mRNA or protein levels of BR2 receptors between young and old animals. BR1 protein levels seemed to be higher in hypertensive animals, however there was no difference between young and old SHR. We have also shown previously that incubation with Ile-Pro-Pro in vitro improves bradykinin-induced arterial vasorelaxation, especially in old vessels, a phenomenon thought to be related to the Ang(1-7)-Mas-receptor axis (14, 24). However, tripeptide feeding had no effects on bradykinin-induced vascular activity in the present study. In addition, pretreatment with the Mas-receptor blocker, A-779, did not exert any effect on bradykinin’s vascular actions, even though Mas receptor expression was increased in young SHRs. An interesting gaseous vasoactive transmitter hydrogen sulphide (H2S) showed, like bradykinin, a biphasic vascular effect, being vasoconstrictor in the low and vasodilator in the high concentrations in aortic preparation in vitro, both in hypertensive and normotensive young rats (33).
In particular in the SHR groups, bradykinin induced vasoconstriction, which was abolished by COX inhibition. There was a trend that especially COX-2 mRNA expression levels were increased in old SHR which may explain the more pronounced vasoconstriction via increased prostaglandin synthesis. However, prostacyclin synthase expression was also increased in old SHR. Tang et al. (34) showed that in old SHR, there is over-expression of prostacyclin synthase and thromboxane synthase in the endothelial cells. However, in that study, only COX-1 mRNA and protein levels were increased in especially in old SHRs compared to aged matched normotensive animals. Based on previous studies (35-37), it seems that bradykinin can induce the release of arachidonic acid (AA) from cell membranes by activating phospholipase A2 (PLA2). Hypertension and ageing increases vascular inflammation (38), COX overexpression and also eNOS expression are decreased in aged hypertensive animals (34), which leads to an imbalance between these constricting and dilating endothelial factors. When bradykinin increases the release of AA, the overexpressed COX converts AA into prostaglandin H2. Due to vascular dysfunction and inflammation, bradykinin induced vasodilatation is transformed into vasoconstriction. Mason et al. (39) showed that diabetic SHR had high nitroxidative stress in aortic endothelium caused by low NO bioavailability and high OONO– (peroxynitrite) levels. It is not known whether bradykinin can influence on endothelial dysfunction by affecting on nitroxidative stress in endothelium. Nitroxidative stress, however, may also be involved bradykinins’ biphasic vascular effects.
ACE-1 activity was elevated in both old hypertensive and old normotensive animals. It has been shown previously that ACE-1 enzyme activity increases with aging (40, 41) but activity in aorta is not affected by hypertension (42). However, controversial results exist: ACE activity is increased in aortal endothelium as well as in the smooth muscle layer of SHR compared to normotensive animals (43). Based on our present data, it seems that the ACE-1 level is not regulated through mRNA expression as it remained unchanged in aorta. Our results from Western blot analysis from the kidney indicated that protein levels of ACE-1 were elevated in the old animals. It may be that at least partly ACE-1 activity is regulated more through protein activation rather than by elevated mRNA expression of the gene.
Interestingly, feeding with ACE-inhibitory peptides did not lower ACE-1 activity in aorta. However, the blood pressure values of the old animals were decreased by peptides. The investigated tripeptides seem to have some other mechanism of action in addition to ACE-1 inhibition, as we have reported before (12-14, 24). In this study, we investigated ACE-1 activity only in aorta. It may be that ACE-1 activity is more extensively altered by ACE-1 inhibitors in other tissues/body fluids such as plasma, kidney, lungs or small arteries.
IL-12, an anti-inflammatory cytokine, levels were highest in normotensive young animals as compared to all other groups. It seems that at least in humans, the IL-12 levels decline with ageing (44). For example, in SHR, IL-12 levels were clearly decreased during ageing. Shaashue et al. (45) showed that stress and surgery lowered IL-12 levels via the prostaglandin pathway. It may be that it was the increased prostaglandin production in aged SHR that decreased the IL-12 levels. Unfortunately, other measured cytokines were below the detection limit of the commercial assay.
To conclude long-term feeding with a drink containing tripeptides lowers or prevents the increase of blood pressure in hypertensive and normotensive old animals. ACE-1 activity, protein levels but not mRNA expression are elevated in old animals. We also demonstrate that vascular inflammation and dysfunction in aged hypertensive animals cause bradykinin to induce vasoconstriction, an effect not modified by tripeptide feeding but apparently related to changes in the prostaglandin pathway.
Acknowledgments: This study was conducted with support from Paivikki and Sakari Sohlberg Foundation, Finnish Clinical Chemistry Foundation and Finska Lakaresallskapet (Einar och Karin Stroems stiftelse), Finland. We thank Dr. Ewen MacDonald for checking the grammar and style of the manuscript.
Conflict of interests: None declared.
REFERENCES
- Jakala P, Vapaatalo H. Antihypertensive peptides from milk proteins. Pharmaceuticals 2010; 3: 251-272.
- Korhonen H, Pihlanto A. Food-derived bioactive peptides - opportunities for designing future foods. Curr Pharm Des 2003; 9: 1297-1308.
- Kris-Etherton PM, Hecker KD, Bonanome A, et al. Bioactive compounds in foods: their role in the prevention of cardiovascular disease and cancer. Am J Med 2002; 113: 71S-88S.
- Fekete A, Givens DI, Lovegrove JA. Casein-derived lactotripeptides reduce systolic and diastolic blood pressure in a meta-analysis of randomised clinical trials. Nutrients 2015; 7: 659-681.
- Jauhiainen T, Niittynen L, Oresic M, et al. Effects of long-term intake of lactotripeptides on cardiovascular risk factors in hypertensive subjects. Eur J Clin Nutr 2012; 66: 843-849.
- Turpeinen AM, Jarvenpaa S, Kautiainen H, Korpela R, Vapaatalo H. Antihypertensive effects of bioactive tripeptides-a random effects meta-analysis. Ann Med 2013; 45: 51-56.
- Xu J, Qin L, Wang P, Li W, Chang C. Effect of milk tripeptides on blood pressure: a meta-analysis of randomized controlled trials. Nutrition 2008; 24: 933-940.
- Ehlers PI, Kivimaki AS, Turpeinen AM, Korpela R, Vapaatalo H. High blood pressure-lowering and vasoprotective effects of milk products in experimental hypertension. Br J Nutr 2011; 106: 1353-1363.
- Sipola M, Finckenberg P, Santisteban J, Korpela R, Vapaatalo H, Nurminen ML. Long-term intake of milk peptides attenuates development of hypertension in spontaneously hypertensive rats. J Physiol Pharmacol 2001; 52: 745-754.
- Ehlers PI, Kivimaki AS, Siltari A, Turpeinen AM, Korpela R, Vapaatalo H. Plant sterols and casein-derived tripeptides attenuate blood pressure increase in spontaneously hypertensive rats. Nutr Res 2017; 32: 292-300.
- Jauhiainen T, Pilvi T, Cheng ZJ, et al. Milk products containing bioactive tripeptides have an antihypertensive effect in double transgenic rats (dTGR) harbouring human renin and human angiotensinogen genes. J Nutr Metab 2010; 2010: 287030. doi: 10.1155/2010/287030
- Lehtinen R, Jauhiainen T, Kankuri E, et al. Effects of milk casein-derived tripeptides Ile-Pro-Pro, Val-Pro-Pro, and Leu-Pro-Pro on enzymes processing vasoactive precursors in vitro. Arzneimittelforschung 2010; 60: 182-185.
- Siltari A, Kivimaki AS, Ehlers PI, Korpela R, Vapaatalo H. Effects of milk casein derived tripeptides on endothelial enzymes in vitro; a study with synthetic tripeptides. Arzneimittelforschung 2012; 62: 477-481.
- Siltari A, Korpela R, Vapaatalo H. Bradykinin -induced vasodilatation: role of age, ACE1-inhibitory peptide, mas- and bradykinin receptors. Peptides 2016; 85: 46-55.
- Hayes M, Stanton C, Slattery H, et al. Casein fermentate of Lactobacillus animalis DPC6134 contains a range of novel propeptide angiotensin-converting enzyme inhibitors. Appl Environ Microbiol 2007; 73: 4658-4667.
- Contreras MdM, Carron R, Montero MJ, Ramos M, Recio I. Novel casein-derived peptides with antihypertensive activity. Int Dairy J 2009; 19: 566-573.
- Regoli D, Nsa Allogho S, Rizzi A, Gobeil FJ. Bradykinin receptors and their antagonists. Eur J Pharmacol 1998; 348: 1-10.
- Feng W, Xu X, Zhao G, et al. Increased age-related cardiac dysfunction in bradykinin B2 receptor-deficient mice. J Gerontol A Biol Sci Med Sci 2016; 71: 178-187.
- Kintsurashvili E, Duka A, Ignjacev I, Pattakos G, Gavras I, Gavras H. Age-related changes of bradykinin B1 and B2 receptors in rat heart. Am J Physiol Heart Circ Physiol 2005; 289: H202-H205.
- Kuoppala A, Shiota N, Lindstedt KA, et al. Expression of bradykinin receptors in the left ventricles of rats with pressure overload hypertrophy and heart failure. J Hypertens 2003; 21: 1729-1736.
- Mantelli L, Amerini S, Ledda F. Bradykinin-induced vasodilation is changed to a vasoconstrictor response in vessels of aged normotensive and hypertensive rats. Inflamm Res 1995; 44: 70-73.
- Nurmi L, Heikkila HM, Vapaatalo H, Kovanen PT, Lindstedt KA. Downregulation of bradykinin type 2 receptor expression in cardiac endothelial cells during senescence. J Vasc Res 2012; 49: 13-23.
- Perez V, Velarde V, Acuna-Castillo C, et al. Increased kinin levels and decreased responsiveness to kinins during aging. J Gerontol A Biol Sci Med Sci 2005; 60: 984-990.
- Ehlers PI, Nurmi L, Turpeinen AM, Korpela R, Vapaatalo H. Casein-derived tripeptide Ile-Pro-Pro improves angiotensin-(1-7)- and bradykinin-induced rat mesenteric artery relaxation. Life Sci 2011; 88: 206-211.
- Claveau D, Sirinyan M, Guay J, et al. Microsomal prostaglandin E synthase-1 is a major terminal synthase that is selectively up-regulated during cyclooxygenase-2-dependent prostaglandin E2 production in the rat adjuvant-induced arthritis model. J Immunol 2003; 170: 4738-4744.
- Sang H, Liu L, Wang L, et al. Opposite roles of bradykinin B1 and B2 receptors during cerebral ischaemia-reperfusion injury in experimental diabetic rats. Eur J Neurosci 2016; 43: 53-65.
- Xue H, Zhou L, Yuan P, et al. Counteraction between angiotensin II and angiotensin-(1-7) via activating angiotensin type I and Mas receptor on rat renal mesangial cells. Regul Pept 2012; 177: 12-20.
- Vandesompele J, De Preter K, Pattyn F, et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 2002; 3(7): RESEARCH0034.
- Schwager SL, Carmona AK, Sturrock ED. A high-throughput fluorimetric assay for angiotensin I-converting enzyme. Nat Protoc 2006; 1: 1961-1964.
- Maestri R, Milia AF, Salis MB, et al. Cardiac hypertrophy and microvascular deficit in kinin B2 receptor knockout mice. Hypertension 2003; 41: 1151-1155.
- Oeseburg H, Iusuf D, van der Harst P, van Gilst WH, Henning RH, Roks AJ. Bradykinin protects against oxidative stress-induced endothelial cell senescence. Hypertension 2009; 53: 417-422.
- Wang T, Wan ZH, Liu JH, et al. Age-related changes in kallikreins-kinins system in rat corpus cavernosum. Int J Androl 2011; 34: 33-40.
- Cacanyiova S, Berenyiova A, Kristek F, Drobna M, Ondrias K, Grman M. The adaptive role of nitric oxide and hydrogen sulphide in vasoactive responses of thoracic aorta is triggered already in young spontaneously hypertensive rats. J Physiol Pharmacol 2016; 67: 501-512.
- Tang EH, Vanhoutte PM. Gene expression changes of prostanoid synthases in endothelial cells and prostanoid receptors in vascular smooth muscle cells caused by aging and hypertension. Physiol Genomics 2008; 32: 409-418.
- Briand SI, Bernier SG, Guillemette G. Calcium-calmodulin plays a major role in bradykinin-induced arachidonic acid release by bovine aortic endothelial cells. J Cell Biochem 1996; 63: 292-301.
- Hecquet C, Biyashev D, Tan F, Erdos EG. Positive cooperativity between the thrombin and bradykinin B2 receptors enhances arachidonic acid release. Am J Physiol Heart Circ Physiol 2006; 290: H948-H958.
- Kennedy CR, Proulx PR, Hebert RL. Role of PLA2, PLC, and PLD in bradykinin-induced release of arachidonic acid in MDCK cells. Am J Physiol 1996; 271: C1064-C1072.
- Rodriguez-Manas L, El-Assar M, Vallejo S, et al. Endothelial dysfunction in aged humans is related with oxidative stress and vascular inflammation. Aging Cell 2009; 8: 226-238.
- Mason RP, Corbalan JJ, Jacob RF, Dawoud H, Malinski T. Atorvastatin enhanced nitric oxide release and reduced blood pressure, nitroxidative stress and rantes levels in hypertensive rats with diabetes. J Physiol Pharmacol 2015; 66: 65-72.
- Arutyunyan TV, Korystova AF, Kublik LN, Levitman MK, Shaposhnikova VV, Korystov YN. Effects of taxifolin on the activity of angiotensin-converting enzyme and reactive oxygen and nitrogen species in the aorta of aging rats and rats treated with the nitric oxide synthase inhibitor and dexamethasone. Age 2012; 35: 2089-2097.
- Emel’yanov M, Korystova A, Kublik L, Levitman M, Shaposhnikova V, Korystov Y. Low doses of ethanol decrease the activity of the angiotensin-converting enzyme in the aorta of aging rats and rats treated with a nitric oxide synthase inhibitor and dexamethasone. Clin Sci (Lond) 2011; 122: 75-81.
- Grima M, Welsch C, Giesen-Crouse EM, Coquard C, Barthelmebs M, Imbs JL. Age-related variations in tissue angiotensin converting enzyme activities: comparison between spontaneously hypertensive and Wistar-Kyoto rats. J Hypertens 1990; 8: 697-702.
- Sim M. Angiotensin converting enzyme in the endothelium and smooth muscle cum adventitia of the normo- and hypertensive rat aorta. Clin Exp Hypertens A 1990; 12: 1107-1113.
- Stefanovic, Golubovic, Vlahovic, Mitic-Zlatkovic. Age-related changes in IL-12 production by peripheral blood mononuclear cells (PBMC). J Intern Med 1998; 243: 83-84.
- Shaashua L, Rosenne E, Neeman E, et al. Plasma IL-12 levels are suppressed in vivo by stress and surgery through endogenous release of glucocorticoids and prostaglandins but not catecholamines or opioids. Psychoneuroendocrinology 2014; 42: 11-23.
A c c e p t e d : May 30, 2017