DUODENOCUTANEOUS FISTULA IN RATS AS A MODEL FOR “WOUND HEALING-THERAPY” IN ULCER HEALING: THE EFFECT OF PENTADECAPEPTIDE BPC 157, L-NITRO-ARGININE METHYL ESTER AND L-ARGININE
INTRODUCTION
We wanted to challenge the understanding of duodenal ulcer disease pathophysiology and therapy, along with Selye’s and Szabo’s cysteamine duodenal lesions, as the mainstay in the duodenal ulcer disease model (1-3), which argues against “wound healing therapy” (i.e., duodenal ulcer heals like the skin ulcer does). This challenge was realized using duodenocutaneous fistulas in rats and the parallel therapy, likely related to nitric oxide (NO)-system, the stable pentadecapeptide BPC 157, (4-9) (an anti-ulcer peptide supposed to be novel mediator of Robert’s cytoprotection (as stable in human gastric juice more than 24 hours)) (4-9), known to heal fistulas (10-12) and skin wounds (13-15), the nitric-oxide synthase (NOS)-substrate L-arginine and/or the NOS-inhibitor L-nitro-arginine methyl ester (L-NAME). Up to now, they were not investigated in parallel, in duodenal ulcers (16, 17), which should be important considering the dual significance of the NO-system (8), and the NO-system’s particular role in skin and gastrointestinal lesions healing (5, 18, 19) as well as fistulas healing (10, 12). Besides, we assume sphincter failure as a hallmark of the ongoing injury, in contrast to some considerations about NO-sphincter relations (20), however, unrelated to injurious conditions (21-23). There, we have to assume the dysfunction of the nitrergic pathway (for instance, excision-immediate heavy loss of endothelium cells from the vascular wall results with less NO-production ability (24)), acting differently on the damaged tissue integrity (10, 25).
In principle, as emphasized before (11), external fistulas form an anomalous connection between skin and gastrointestinal tract that provides a direct and unusual contact between these different, normally separated, tissues, thereby resulting in special new circumstances and healing difficulties, and a failure of regular healing that is otherwise not present. In particular, this fistula is the only new healing model that practically combines the potentially different processes that might occur simultaneously at the skin and other gastrointestinal wound, and provide insight into the special healing effects of agents under investigation (i.e., the skin wound healing effect could contribute to the healing of gastrointestinal ulcer and vice versa, while spontaneous healing is lacking). Fistula closing may therefore be a useful self-controlling system for both skin and gastrointestinal defect healing (10-12). We should emphasize that BPC 157 (as described (4-9)) avoids all the practical pitfalls for fistula healing using peptidergic growth factors (i.e., standard growth factors would hardly combine skin and gastrointestinal ulcer healing due to the different way of administration and drug delivery to gastrointestinal ulcer and skin ulcer (26, 27)). Of note, BPC 157 is always given alone, and thereby may act directly, always using the same dosage range. This is different from the commonly used standard growth factors (i.e., EGF) which are given with carriers (4-9) and require special delivery systems for skin healing (but not for gastrointestinal ulcer healing) (26, 27).
Therefore, in view of the current reports (4-12), we could envisage a duodenocutaneous fistula model with the BPC 157 application, using the same dose, with skin healing as part of the corresponding healing of gastrointestinal, and in particular, duodenal ulcer healing.
Once the rat duodenocutaneous fistulas are resolved, the consequences thereof could be over the present insignificance of duodenocutaneous fistulas (i.e., a very rare complication of duodenal ulcer disease, most often felt to occur following ulcer surgery (28), but experimentally not investigated), or the possibility of the skin wound that would constitute an entirely different type of the tissue than the gut mucosa and subepithelial tissues unreliable for common therapy.
Finally, in view of several recent reports (4-12), orderly development of rat fistulas (made by direct sutured anastomosis between the two structures (10-12)), fistulas suddenly developed, relentlessly progressive (even for a period of several weeks or more, if not lethal) (10-12) might accordingly reproduce the phenomenon of poor fistulas healing which may occur during spontaneous disease.
Thus, we could suggest that the resolved rat duodenocutaneous fistulas, the newly definable course of the duodenal ulcer (that we initially controlled) as like that of the skin ulcer (that also we initially controlled). The anatomic (duodenal defect at ventral side, 2 mm from pylorus) and clinical identity (poor and long lasting healing) of what we now call duodenocutaneous fistula would ascertain a complicated duodenal and skin ulcer and their implemented simultaneous healing (with a suited therapy) or the persistent partial (one defect left) or complete (both defects left) failure (without adequate therapy).
This mandates that a successful BPC 157 effect (previously, without acting on gastric acid secretion (4-9), it healed the cysteamine pathology (29-33)), should be the synchronized healing of the different tissues, as shown with the healing of the gastrocutaneous, esophagocutaneous, and colocutaneous fistulas, also through the NO-system (10-12), and also interacting with NO-agents in different models and species (10, 12, 25, 34-40). Likely, this could be the extended NO-system involvement (with the gastric lesions, despite inconsistencies, L-arginine has a beneficial effect, while L-NAME has an ulcerogenic effect (8, 36)), they were far less investigated in duodenal lesions (16, 17).
Indeed, BPC 157 has a particular potential to act as combining therapy in the whole gastrointestinal tract (5, 6) (esophageal (41-43), gastric (31, 36, 44), duodenal (29-33) and colitis lesions’ counteraction (12)), and the ameliorated course after the experimental creation of the more complex conditions and complications (44-46) in rats, including those induced by various NSAIDs (47-49). Besides, BPC 157 has particular effects on the wound-healing process in different tissues (50-55), including blood vessels (9, 30, 39, 52-54) and in particular burn wounds (13-15). This is most likely due to the stimulation of the early growth response-1 (egr-1) gene and its co-repressor nerve growth factor 1-A binding protein-2 (naB2), which are also responsible for cytokine and growth factor generation and thereby, early extracellular matrix (collagen) and blood vessel formation (55). The others correlated the BPC 157 beneficial effects with the activation of a cellular focal adhesion kinase (FAK)-paxillin signal pathway and subsequently demonstrated that BPC 157, dose- and time-dependently, increased the expression of growth hormone receptor, Janus kinase 2 (JAK-2), the downstream signal pathway of growth hormone receptor (56, 57).
Thus, responding to this novel model challenge with rat duodenocutaneous fistula, we suggest an additional particular wound healing background (13-15, 54, 57, 58) for fistula healing (10-12) and potential agent’s efficacy.
For practical purposes, the stable gastric pentadecapeptide BPC 157 was given daily, intraperitoneally or perorally, in drinking water, using the previous efficacious regimens as in the other external fistulas healing trials (10-12). In addition, these effects (stable gastric pentadecapeptide BPC 157, along with NOS-blockade, L-NAME, and NOS-substrate L-arginine application (5)), might better clarify the healing of duodenal and skin defects and duodenocutaneous fistulas, and find possible simultaneous or separate healing, as a new NO-system related phenomenon.
MATERIALS AND METHODS
Animals
Wistar Albino male rats (200 g b.w.) were randomly assigned to the experiments (10 animals, at least, per each experimental group and interval), all of which were approved by the Local Ethics Committee. Furthermore, all experiments were carried out under blind protocol and the effect was assessed by examiners who were completely unaware of the given protocol.
Drugs
Pentadecapeptide BPC 157 (GEPPPGKPADDAGLV, M.W. 1419), (Diagen, Ljubljana, Slovenia) dissolved in saline, was used in all experiments. The peptide, BPC 157, is part of the sequence of human gastric juice protein, BPC, and is freely soluble in water at pH 7.0 and saline. It was prepared as described previously with 99% high pressure liquid chromatography (HPLC) purity, expressing 1-des-Gly peptide as an impurity (4-12, 30). L-NAME (Sigma, USA) and L-arginine (Sigma, USA) were accordingly used.
Procedure
Using a previous procedure, the direct anastomosis between the two structures (10-12), in deeply anaesthetized rats. The purpose of surgery was the creation of a duodenocutaneous fistula (2 mm diameter duodenum (at ventral side, 2 mm from pylorus) and 3 mm diameter skin defect at the proximal third part of medial incision, where a precise caliper was used to verify the initial size of the defect), as described before (11). Direct anastomosis between the two structures, duodenum and skin, was performed and sutured (Vycril 2-0, Johnson & Johnson) to form a channel.
Experimental protocol
Using a previous procedure and regimens (10-12), BPC 157 was given perorally, in drinking water (10 µg/kg, 10 ng/kg, i.e., 0.16 µg/mL, 0.16 ng/mL, 12 mL/rat/day) till sacrifice, or alternatively, 10 µg/kg, 10 ng/kg intraperitoneally, first application at 30 min after surgery, last at 24 hours before sacrifice. L-NAME (5 mg/kg intraperitoneally) and/or L-arginine (100 mg/kg intraperitoneally) were given alone or together, first application at 30 min after surgery, last at 24 hours before sacrifice. BPC 157 10 µg/kg, intraperitoneally or perorally, was given with L-NAME (5 mg/kg intraperitoneally) and/or L-arginine (100 mg/kg intraperitoneally).
Controls simultaneously received an equivolume of saline (5.0 mL/kg intraperitoneally) or water only.
The assessment was at day 1, 2, 3, 7, 14 and 21 as follows.
Duodenum defect, skin defect, fistula assessment
A precise caliper was used to verify the final size of the defect. The largest diameter of the skin or duodenal defect was assessed (mm), photographed and further verified using the program ISSA (VAMSTEC Software Company, Zagreb, Croatia), and the tissue processed for further microscopic analysis (10-12).
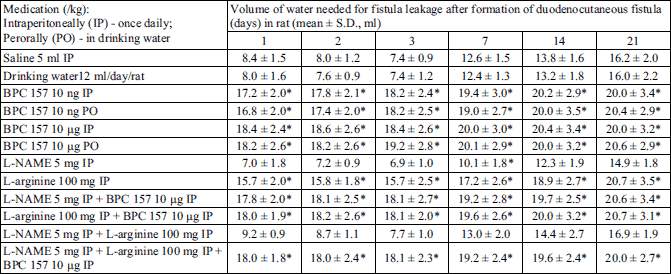
To assess fistula leakage, a separate group of animals received a volume of water intragastrically (1 ml/6 s), either to leaking induction, or to a maximal volume of 20 ml (10-12). In this case, if leakage did not occur after 5 min, the fistula was considered functionally closed.
Lower esophageal sphincter pressure assessment and pyloric sphincter pressure assessment
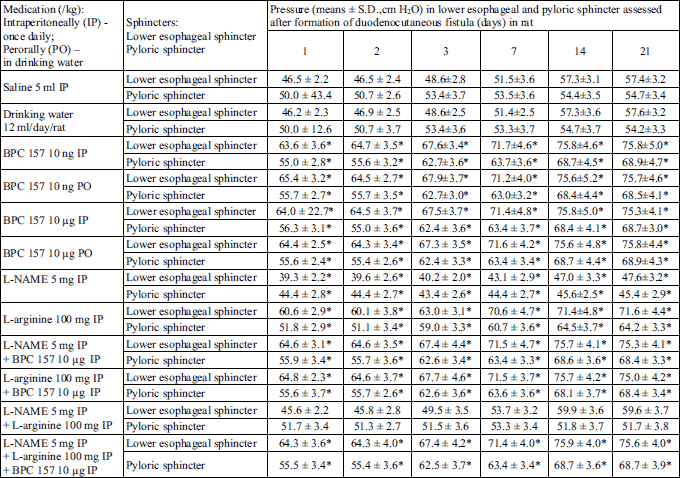
As described before (10, 25, 41-43), in all rats, manometrical evaluation (cm H2O) was performed with a water manometer connected to the drainage port of the Foley catheter as described (the values of 68 – 76 cm H2O for lower esophageal sphincter, and 68 – 74 cm H2O for pyloric sphincter, were considered to be normal as determined before (10, 25, 41-43)). The proximal side of the esophageal, or distal side of the duodenal incision, was ligated to prevent regurgitation.
Mortality assessment
Mortality rate (%) was assessed throughout the study (45).
Statistical analyses
Statistical analysis was performed by a non-parametric Kruskal-Wallis ANOVA, and subsequent Mann-Whitney U-test, to compare groups. Fisher’s exact probability test for mortality rate assessment was used. Values of P < 0.05 were considered statistically significant.
RESULTS
We demonstrated a consistent counteracting beneficial effect in all BPC 157 rats (i.e., given perorally (in drinking water) or intraperitoneally), on its own, and with NOS-blockade (L-NAME) and/or NOS-substrate (L-arginine). A simultaneous effect was demonstrated on the closure of skin, duodenal, fistula defects; healing occurred macro/microscopically, biomechanically, and functionally, as well as reinstating sphincter functions; no mortality. On the other hand, an opposite persistent lesion course was portrayed in controls (persistent lesions, leaking, failure biomechanically and functionally, low pressure within sphincters, and mortality). There was a counteracting beneficial effect of L-arginine, and an opposite, additionally aggravating one of L-NAME, with their mutual counteraction. Thus, in rats’ duodenocutaneous fistulas the duodenal and the skin defect follow the same pattern of healing and therapy (Tables 1-5, Fig. 1).
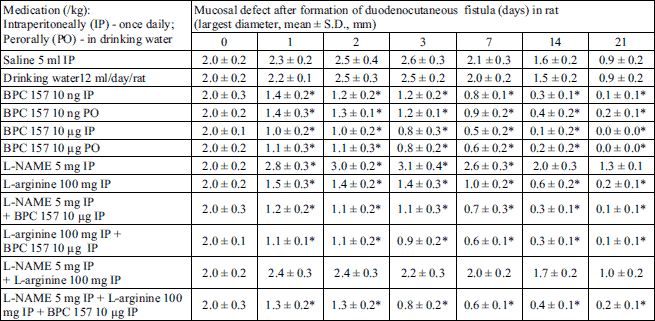
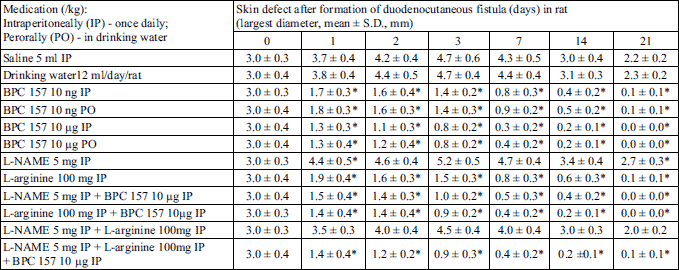
As mentioned, if not treated otherwise, duodenocutaneous fistulas exhibited poor healing (Tables 1-5, Fig. 1), with persistence of defects (Tables 1 and 2), continuous fistula leakage (Table 3), sphincter failure (Table 4) and significant mortality rate (Table 5). BPC 157 given perorally or intraperitoneally, in µg- and ng-regimens, rapidly improved complete presentation (Tables 1-5, Fig. 1) (maximal volume instilled already at 7th day (Table 3)). Considering the NO-agents effects, and NO-system involvement, L-arginine was also beneficial in all aspects (no mortality; however, maximal volume instilled not before 21th day) (Tables 1-5) while L-NAME application further aggravated entire duodenocutaneous fistulas course (Tables 1-5), with even higher mortality rate (Table 5). Considering the possible mutual interaction, L-arginine brings down aggravation by NOS-blockade with L-NAME to the control levels. Interestingly, L-NAME appears to be more sensitive to BPC 157 application providing that BPC 157 more than nullifies the effect of L-NAME (i.e., L-NAME + BPC 157; L-NAME + L-arginine + BPC 157 markedly improved over control values) (Tables 1-5). Thus, BPC 157 effects appear to be stronger than those of L-arginine, and this is illustrated in the earlier biomechanical improvement, in the early interval, where BPC 157 + L-arginine rats achieved the maximal volume.
Generally, in duodenocutaneous fistula-rats (Fig. 1) the microscopic presentation (i.e., severe necrosis along the fistula wall, including a large necrotic area of superficial epithelium and a broad band of necrotic subcutaneous tissue and muscle after 3 days, fistulous channel walled with granulation tissue after 14 days) followed the described macroscopical healing and previous course of gastrocutaneous fistula-rats (11) (initially, in animals treated with BPC 157, the edema was less pronounced, the superficial epithelium surrounding the defect was practically intact, and the defect was closed by crust, and after 14 days, unlike in the controls, the treated animals showed poor granulation tissue formation and prominent regeneration of subcutaneous muscle fibers) (11).
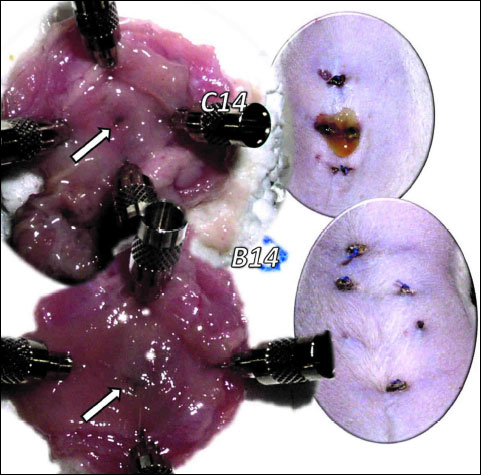 |
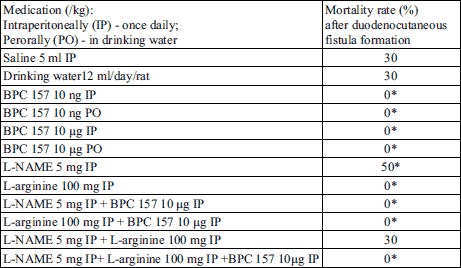
Fig. 1. Presentation of the duodenal and cutaneous defect at the 14th day after duodenocutaneous fistula formation in control (C14) and BPC 157 (10 µg/kg i.p., once daily) (B14) treated rats. Note in controls a still open duodenal defect (arrow) and apparent leaking through the skin fistula defect while BPC 157 rats presented with already closed both duodenal (arrow) and skin defects and no fistula leaking at all. |
DISCUSSION
With rats’ duodenocutaneous fistula, the anti-ulcer peptide stable gastric pentadecapeptide BPC 157 (4-12), L-arginine and L-NAME, we considered the “wound healing therapy” notion, versus the conventional understanding of duodenal disease and therapy and the mainstream cysteamine duodenal ulcer model (1-3).
We demonstrated that the rats’ duodenocutaneous fistula healing model, methodologically and therapeutically (BPC 157, L-arginine), strongly combines the healing and therapy of the duodenal and skin defects, NO-system involvement (i.e., counteraction of L-NAME (as harmful effect of NOS-blockade)). Thus, we revealed the essential healing commonality between the BPC 157, L-arginine, L-NAME and NO-system (10, 12); we assumed, due to the injurious tissue loss (24), initial NO-system negative dysfunction that had been manipulated by BPC 157, L-arginine and L-NAME application manipulating in both ways, before tissue injury NO-essential background improved or further aggravated.
Furthermore, in methodology, the controlled (dis)advantages (the uniformly formed defects of the skin and duodenum, then, the persistent lesions, leaking, failure biomechanical and functional, low pressure within sphincters, a long-standing course), causally related, explain that in rat duodenocutaneous fistulas more lesions lead to less sphincter pressure (10, 25, 41-43). Therefore, L-NAME further decreased pressure and L-arginine increased pressure, depending on their negative or positive healing effect. These are in accordance with the effects noted in the esophagocutaneous fistulas- or hyperkalemia-sphincter dysfunction (10, 25) and the obtained sphincters’ interrelation (i.e., the failure of the pyloric sphincter induced the failure of the lower esophageal sphincter (41-43)). Thus, these are in seeming contrast to some considerations about NO-sphincter relations (20-23), providing the dysfunction of the nitrergic pathway differently acting at the damaged tissue integrity (and obviously prevailed and different from those of NO-esophageal sphincter relaxation (20), obtained in intact dogs (21) or ferrets (22), or muscle strips (23)).
Likely, this NO-system negative dysfunction could be a submaximal one as suggested before (36); since it could be clearly improved (BPC 157, L-arginine) while more hardly aggravated by an additional NOS-blockade (note, L-NAME administration effect is also not significantly different than that of saline control and drinking water at some of respective times). Commonly, BPC 157 counteracted L-NAME better than L-arginine did (8, 10, 12, 25, 34-40), (L-NAME-worsening was counteracted with the L-arginine effect and vice versa (L-NAME + L-arginine brought to the level of the control), while BPC 157 more than negated the L-NAME-worsening (L-NAME + L-arginine and L-NAME + BPC 157; L-NAME + L-arginine + BPC 157 brought below the level of the control), and accordingly, BPC 157 induces NO-release from gastric mucosa supernatant, like L-arginine, but also in conditions where L-arginine is not working (36).
Consequently, the accurate attribution of the obtained peptide effects, particularly for skin defect healing (where unlike uncertain healing attribution of other peptides which are given with carriers (4-9), BPC 157 is given alone and thereby acts directly), the therapeutic success of BPC 157 over the external fistulas model (10-12) and now in duodenocutaneous fistulas further emphasized the real healing congruence of the skin healing and other gastrointestinal tissues healing. For the healing congruence of both defects, it might be possible that the skin healing methodologically resolves the therapy phenomenon in other gastrointestinal tissues or the skin healing was affected by the healing of any part of the gastrointestinal tract that was connected to the skin. Using the same dose range, BPC 157 achieves the requested healing congruence. Illustratively, BPC 157 had accelerated the healing of severe burns, topically as a cream, or systemically (13-15); BPC 157 had counteracted the severe healing impairment otherwise induced with systemic corticosteroid application (13-15), or alloxan-hyperglicemia (58), overpowered the skin wound healing of the corresponding standard agents (i.e., becaplermin, recombinant human platelet-derived growth factor homodimer of B chains, PDGF-BB; silver sulfadiazine cream) (13-15, 55, 58, 59). In the used dose range this correlated with the counteraction of the esophageal (41-43), gastric (31, 36, 44), duodenal (29-33) and colonic mucosal lesions (12), mitigated and ameliorated the course after an experimental creation of the more complex conditions and complications in rats (44-46), and counteracted complex pathology induced by various NSAIDs (47-49). In continuation, this correlates with the healing of created external fistulas (10-12), simultaneous healing of esophageal, gastric, colonic and skin defects (10-12), and now also, the duodenal defect. Thus, considering the effectiveness after peroral administration, it may be possible that this original anti-ulcer peptide, stable and present in human gastric juice, supposed to maintain mucosal integrity (4-9, 29) (BPC 157 is claimed to be a novel mediator of Robert’s cytoprotection (4)), may also benefit the skin part of the fistula defect and exert there its beneficial effect. It may be, considering the effectiveness of BPC 157 after intraperitoneal administration, that these are the NO mediating the skin C-fiber reflexes, that mediate skin vasodilatation (60), providing that BPC 157 maintains C-fibres integrity in both neonatal and adults rats that received capsaicin (61).
In this, the practical peptidergic activity is seen by the fact that the peroral (in drinking water) application corresponds to that of the parenteral administration (4-9, 29). Likely important for the fistula’s healing as well, is the particular cytoprotective background (4). More advanced cytoprotective effects for more tissue healing show it to be a particular wound-healing mediator as well (4, 62), and also supported by advanced healing of severe skin wounds (13-15). Using 3H-labelled pentadecapeptide BPC 157, t/2 was 66 h and 69 h in male and female rats after single oral administration (4-9). In addition, pentadecapeptide BPC 157’s beneficial effect (both in wound and mucosa healing) involves (4-9) reduction of the inflammatory cell numbers, leukotriene B4 (LTB4), thromboxane B2 (TXB2), and myeloperoxidase (MPO) levels in the serum and inflamed tissues (63, 64), and it also increases the macrophage activity (65).
Finally, although this was not especially investigated in the present study, whether this is a receptor-mediated action (57) or not (further studies are required to finalize all these findings (4-9)), they have to be specially related to its molecular effects (55-57). For instance, prominently stimulated expression of the egr-1 gene includes cytokine and growth factor generation, and early extracellular matrix (collagen) formation, and blood vessel function and its repressor naB2 (55), very likely provides a particular feedback-process consistently responsible for the simultaneous healing of two different tissues and fistulas healing (10-12). For example, in duodenal ulcers, the increase in the expression of egr-1 is followed by upregulation of angiogenic growth factors (e.g., VEGF, bFGF, PDGF) (66).
Of note, these additional healing mechanisms of BPC 157, besides those with NO-system (8), show that advanced healing (and collagen) processes (55) fairly reflect the biomechanical improvement (13-15, 45, 46), and vice versa. Thereby, the biomechanical improvement in all BPC 157-rats sustained the markedly advanced volume challenge before leakage, quickly progressing until the successful instillation of the maximal volume, which was achieved already at very early intervals. This indicates the rapidly salvaged fistulas and the advanced healing processes in all BPC 157-rats over the completely failed healing and the consequently small amount of volume that could be sustained in the controls. Also, the advanced biomechanical healing was common in various wounds (13-15, 50, 51, 59), fistulas (10-12), and revealed in the remaining intestine function recovery (45, 46), in BPC 157-studies. Also, based on the observed L-arginine (maximal volume sustained quite later, only at the latest interval; note, that maximal volume was achieved earlier in BPC 157 + L-arginine-rats and BPC 157 + L-arginine + L-NAME-rats) and L-NAME effects, these occurrences are obviously related to the NO-system as well.
BPC 157 also has a strong direct angiogenic healing potential (13-15, 50-52, 59) (a particular one since it is present along with the healing processes in hypocellular and hypovascular tissues (13-15, 50-52, 59), but not in vitro conditions (52)). The direct endothelium protection (4, 8, 31), could partly explain its mentioned influence on the NO-system (8), with a counteracted overexpression of endothelin (38), (note, endothelin has potent ulcerogenic and vasoconstrictor actions in the stomach (67), counteraction of the effects of NOS-inhibitors and NO-precursor (8, 10, 12, 25, 34-40), effects which are seen in particular with external fistula healing (10, 12).
Besides, considering the complexity of the described healing, we should assume that additional mechanisms might be also involved (68, 69).
In conclusion, this study revealed the success of the stable gastric pentadecapeptide, BPC 157, in the therapy of duodenocutaneous fistulas in rats, in a well-controlled model equalizing duodenal and skin ulcer healing, with sphincter function reinstated. It has also shown a likely practical, cytoprotective significant effect, in duodenal ulcer and disease, applicable as a further wound healing therapy in fistulas along with the beneficial effect on other fistula healing (10-12) that should be more than experimentally relevant providing its initial use in the ulcerative colitis trials and it’s very safe profile (4-9, 29).
Summarizing, BPC 157, given perorally, or parenterally, promptly improved healing of both duodenal and skin lesions and mediated fistula closing (macro-/microscopically, and functionally, with no fistula leakage upon application of the maximal water volume, and reinstated the sphincter function). This effect was shared by L-arginine, with an opposite effect seen by L-NAME, thereby portraying NO-system involvement in the healing of duodenal and skin defects, duodenocutaneous fistula closing and reinstated sphincter functions. Hopefully, by resolving the duodenocutanenous fistula-healing, and NO-system involvement, this should illustrate a further wound healing therapy to heal duodenal ulcers.
This article is based on Authors presentations at the 1st Global Gastrointestinal Club of Experimental Biology/IUPHAR GI Section of ASPET “The Best of my Research: The Last 10 Years” meeting organized in Boston 2013 by Prof. Sandor Szabo, University of California, Irvine and GI Section VA Medical Center, Long Beach, California USA.
Conflict of interests: None declared.
REFERENCES
- Selye H, Szabo S. Experimental model for production of perforating duodenal ulcers by cysteamine in the rat. Nature 1973; 244: 458-459.
- Szabo S. Hans Selye and the development of the stress concept. Special reference to gastroduodenal ulcerogenesis. Ann NY Acad Sci 1998; 851: 19-27.
- Szabo S. Dopamine disorder in duodenal ulceration. Lancet 1979; 2: 880-882.
- Sikiric P, Seiwerth S, Brcic L, et al. Revised Robert’s cytoprotection and adaptive cytoprotection and stable gastric pentadecapeptide BPC 157. Possible significance and implications for novel mediator. Curr Pharm Des 2010; 16: 1224-1234.
- Sikiric P, Seiwerth S, Rucman R, et al. Stable gastric pentadecapeptide BPC 157: novel therapy in gastrointestinal tract. Curr Pharm Des 2011; 17: 1612-1632.
- Sikiric P, Seiwerth S, Rucman R, et al. Focus on ulcerative colitis: stable gastric pentadecapeptide BPC 157. Curr Med Chem 2012; 19: 126-132.
- Sikiric P, Seiwerth S, Rucman R, et al. Toxicity by NSAIDs. Counteraction by stable gastric pentadecapeptide BPC 157. Curr Pharm Des 2013; 19: 76-83.
- Sikiric P, Seiwerth S, Rucman R, et al. Stable gastric pentadecapeptide BPC 157 - NO-system relation. Curr Pharm Des 2014; 20: 1126-1135.
- Seiwerth S, Brcic L, Vuletic LB, et al. BPC 157 and blood vessels. Curr Pharm Des 2014; 20: 1121-1125.
- Cesarec V, Becejac T, Misic M, et al. Pentadecapeptide BPC 157 and the esophagocutaneous fistula healing therapy. Eur J Pharmacol 2013; 701: 203-212.
- Skorjanec S, Dolovski Z, Kocman I, et al. Therapy for unhealed gastrocutaneous fistulas in rats as a model for analogous healing of persistent skin wounds and persistent gastric ulcers: stable gastric pentadecapeptide BPC 157, atropine, ranitidine, and omeprazole. Dig Dis Sci 2009; 54: 46-56.
- Klicek R, Sever M, Radic B, et al. Pentadecapeptide BPC 157, in clinical trials as a therapy for inflammatory bowel disease (PL14736), is effective in the healing of colocutaneous fistulas in rats: role of the nitric oxide-system. J Pharmacol Sci 2008; 108: 7-17.
- Bilic M, Bumber Z, Blagaic AB, Batelja L, Seiwerth S, Sikiric P. The stable gastric pentadecapeptide BPC 157, given locally, improves CO2 laser healing in mice. Burns 2005; 31: 310-315.
- Sikiric P, Seiwerth S, Mise S, et al. Corticosteroid-impairment of healing and gastric pentadecapeptide BPC-157 creams in burned mice. Burns 2003; 29: 323-334.
- Mikus D, Sikiric P, Seiwerth S, et al. Pentadecapeptide BPC 157 cream improves burn-wound healing and attenuates burn-gastric lesions in mice. Burns 2001; 27: 817-827.
- Araki H, Komoike Y, Matsumoto M, Tanaka A, Takeuchi K. Healing of duodenal ulcers is not impaired by indomethacin or rofecoxib, the selective COX-2 inhibitor, in rats. Digestion 2002; 66: 145-153.
- Takeuchi K, Suzuki K, Araki H, Mizoguchi H, Sugamoto S, Umdeda M. Roles of endogenous prostaglandins and nitric oxide in gastroduodenal ulcerogenic responses induced in rats by hypothermic stress. J Physiol (Paris) 1999; 93: 423-431.
- Spallotta F, Cencioni C, Straino S, et al. A nitric oxide-dependent cross-talk between class I and III histone deacetylases accelerates skin repair. J Biol Chem 2013; 288: 11004-11012.
- Moncada S, Palmer RM, Higgs EA. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev 1991; 43: 109-142.
- Sidhu AS, Triadafilopoulos G. Neuro-regulation of lower esophageal sphincter function as treatment for gastroesophageal reflux disease. World J Gastroenterol 2008; 14: 985-990.
- Sanmiguel CP, Hagiike M, Mintchev MP, et al. Effect of electrical stimulation of the LES on LES pressure in a canine model. Am J Physiol Gastrointest Liver Physiol 2008; 295: G389-G394.
- Niedringhaus M, Jackson PG, Evans SR, Verbalis JG, Gillis RA, Sahibzada N. Dorsal motor nucleus of the vagus: a site for evoking simultaneous changes in crural diaphragm activity, lower esophageal sphincter pressure, and fundus tone. Am J Physiol Regul Integr Comp Physiol 2008; 294: R121-R131.
- Braverman AS, Vegesna AK, Miller LS, et al. Pharmacologic specificity of nicotinic receptor-mediated relaxation of muscarinic receptor precontracted human gastric clasp and sling muscle fibers within the gastroesophageal junction. J Pharmacol Exp Ther 2011; 338: 37-46.
- Berra-Romani R, Avelino-Cruz JE, Raqeeb A, et al.Ca2+-dependent nitric oxide release in the injured endothelium of excised rat aorta: a promising mechanism applying in vascular prosthetic devices in aging patients. BMC Surg 2013; 13 (Suppl. 2): S40.
- Barisic I, Balenovic D, Klicek R, et al. Mortal hyperkalemia disturbances in rats are NO-system related. The life saving effect of pentadecapeptide BPC 157. Regul Pept 2013; 181: 50-66.
- Brzozowski T, Konturek PC, Konturek SJ, et al. Effect of local application of growth factors on gastric ulcer healing and mucosal expression of cyclooxygenase-1 and -2. Digestion 2001; 64: 15-29.
- Banerjee I, Mishra D, Das T, Maiti TK. Wound pH-responsive sustained release of therapeutics from a poly(NIPAAm-co-AAc) hydrogel. J Biomater Sci Polym Ed 2012; 23: 111-132.
- Reddy AN. Spontaneous duodenocutaneous fistula: a rare complication of duodenal ulcer. Postgrad Med J 1988; 64: 328-330.
- Klicek R, Kolenc D, Suran J, et al. Stable gastric pentadecapeptide BPC 157 heals cysteamine-colitis and colon-colon-anastomosis and counteracts cuprizone brain injuries and motor disability. J Physiol Pharmacol 2013; 64: 597-612.
- Sikiric P, Mikus D, Seiwerth S, et al. Pentadecapeptide BPC 157, cimetidine, ranitidine, bromocriptine, and atropine effect in cysteamine lesions in totally gastrectromized rats: a model for cytoprotective studies. Dig Dis Sci 1997; 42: 1029-1037.
- Sikiric P, Seiwerth S, Grabarevic Z, et al. The beneficial effect of BPC 157, a 15 amino acid peptide BPC fragment, on gastric and duodenal lesions induced by restraint stress, cysteamine and 96% ethanol in rats. A comparative study with H2 receptor antagonists, dopamine promotors and gut peptides. Life Sci 1994; 54: PL63-PL68.
- Bedekovic V, Mise S, Anic T, et al. Different effect of antiulcer agents on rat cysteamine-induced duodenal ulcer after sialoadenectomy, but not gastrectomy. Eur J Pharmacol 2003; 477: 73-80.
- Sikiric P, Seiwerth S, Grabarevic Z, et al. Cysteamine-colon and cysteamine-duodenum lesions in rats. Attenuation by gastric pentadecapeptide BPC 157, cimetidine, ranitidine, atropine, omeprazole, sulphasalazine and methylprednisolone. J Physiol (Paris) 2001; 95: 261-270.
- Balenovic D, Bencic ML, Udovicic M, et al. Inhibition of methyldigoxin-induced arrhythmias by pentadecapeptide BPC 157: a relation with NO-system. Regul Pept 2009; 156: 83-89.
- Boban-Blagaic A, Blagaic V, Romic Z, et al. The influence of gastric pentadecapeptide BPC 157 on acute and chronic ethanol administration in mice. The effect of N(G)-nitro-L-arginine methyl ester and L-arginine. Med Sci Monit 2006; 12: BR36-BR45.
- Sikiric P, Seiwerth S, Grabarevic Z, et al. The influence of a novel pentadecapeptide, BPC 157, on N(G)-nitro-L-arginine methylester and L-arginine effects on stomach mucosa integrity and blood pressure. Eur J Pharmacol 1997; 332: 23-33.
- Grabarevic Z, Tisljar M, Artukovic B, et al. The influence of BPC 157 on nitric oxide agonist and antagonist induced lesions in broiler chicks. J Physiol (Paris) 1997; 91: 139-149.
- Lovric-Bencic M, Sikiric P, Hanzevacki JS, et al. Doxorubicine-congestive heart failure-increased big endothelin-1 plasma concentration: reversal by amlodipine, losartan, and gastric pentadecapeptide BPC157 in rat and mouse. J Pharmacol Sci 2004; 95: 19-26.
- Balenovic D, Barisic I, Prkacin I, et al. Mortal furosemide-hypokalemia-disturbances in rats NO-system related. Shorten survival by L-NAME. Therapy benefit with BPC 157 more than with L-arginine. J Clin Exp Cardiol 2012; 3: 201.
- Stupnisek M, Kokot A, Drmic D, et al. Pentadecapeptide BPC 157 reduces bleeding and thrombocytopenia after amputation in rats treated with heparin, warfarin, L-NAME and L-arginine. PLoS One 2015; 10: e0123454.
- Petrovic I, Dobric I, Drvis P, et al. An experimental model of prolonged esophagitis with sphincter failure in the rat and the therapeutic potential of gastric pentadecapeptide BPC 157. J Pharmacol Sci 2006; 102: 269-277.
- Dobric I, Drvis P, Petrovic I, et al. Prolonged esophagitis after primary dysfunction of the pyloric sphincter in the rat and therapeutic potential of the gastric pentadecapeptide BPC 157. J Pharmacol Sci 2007; 104: 7-18.
- Petrovic I, Dobric I, Drmic D, et al. BPC 157 therapy to detriment sphincters failure-esophagitis-pancreatitis in rat and acute pancreatitis patients low sphincters pressure. J Physiol Pharmacol 2011; 62: 527-534.
- Ilic S, Brcic I, Mester M, et al. Over-dose insulin and stable gastric pentadecapeptide BPC 157. Attenuated gastric ulcers, seizures, brain lesions, hepatomegaly, fatty liver, breakdown of liver glycogen, profound hypoglycemia and calcification in rats. J Physiol Pharmacol 2009; 60 (Suppl. 7): 107-114.
- Vuksic T, Zoricic I, Brcic L, et al. Stable gastric pentadecapeptide BPC 157 in trials for inflammatory bowel disease (PL-10, PLD-116, PL14736, Pliva, Croatia) heals ileoileal anastomosis in the rat. Surg Today 2007; 37: 768-777.
- Sever M, Klicek R, Radic B, et al. Gastric pentadecapeptide BPC 157 and short bowel syndrome in rats. Dig Dis Sci 2009; 54: 2070-2083.
- Ilic S, Drmic D, Zarkovic K, et al. High hepatotoxic dose of paracetamol produces generalized convulsions and brain damage in rats. A counteraction with the stable gastric pentadecapeptide BPC 157 (PL 14736). J Physiol Pharmacol 2010; 61: 241-250.
- Ilic S, Drmic D, Zarkovic K, et al. Ibuprofen hepatic encephalopathy, hepatomegaly, gastric lesion and gastric pentadecapeptide BPC 157 in rats. Eur J Pharmacol 2011; 667: 322-329.
- Ilic S, Drmic D, Franjic S, et al. Pentadecapeptide BPC 157 and its effects on a NSAID toxicity model: diclofenac-induced gastrointestinal, liver, and encephalopathy lesions. Life Sci 2011; 88: 535-542.
- Staresinic M, Petrovic I, Novinscak T, et al. Effective therapy of transected quadriceps muscle in rat: gastric pentadecapeptide BPC 157. J Orthop Res 2006; 24: 1109-1117.
- Staresinic M, Sebecic B, Patrlj L, et al. Gastric pentadecapeptide BPC 157 accelerates healing of transected rat Achilles tendon and in vitro stimulates tendocytes growth. J Orthop Res 2003; 21: 976-983.
- Brcic L, Brcic I, Staresinic M, Novinscak T, Sikiric P, Seiwerth S. Modulatory effect of gastric pentadecapeptide BPC 157 on angiogenesis in muscle and tendon healing. J Physiol Pharmacol 2009; 60 (Suppl. 7): 191-196.
- Hrelec M, Klicek R, Brcic L, et al. Abdominal aorta anastomosis in rats and stable gastric pentadecapeptide BPC 157, prophylaxis and therapy. J Physiol Pharmacol 2009; 60 (Suppl. 7): 161-165.
- Stupnisek M, Franjic S, Drmic D, et al. Pentadecapeptide BPC 157 reduces bleeding time and thrombocytopenia after amputation in rats treated with heparin, warfarin or aspirin. Thromb Res 2012; 129: 652-659.
- Tkalcevic VI, Cuzic S, Brajsa K, et al. Enhancement by PL 14736 of granulation and collagen organization in healing wounds and the potential role of egr-1 expression. Eur J Pharmacol 2007; 570: 212-221.
- Chang CH, Tsai WC, Lin MS, Hsu YH, Pang JH. The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration. J Appl Physiol 2011; 110: 774-780.
- Chang CH, Tsai WC, Hsu YH, Pang JH. Pentadecapeptide BPC 157 enhances the growth hormone receptor expression in tendon fibroblasts. Molecules 2014; 19: 19066-19077.
- Seveljevic-Jaran D, Cuzic S, Dominis-Kramaric M, et al. Accelerated healing of excisional skin wounds by PL 14736 in alloxan-hyperglycemic rats. Skin Pharmacol Physiol 2006; 19: 266-274.
- Seiwerth S, Sikiric P, Grabarevic Z, et al. BPC 157’s effect on healing. J Physiol (Paris) 1997; 91: 173-178.
- Boutsiouki P, Georgiou S, Clough GF. Recovery of nitric oxide from acetylcholine-mediated vasodilatation in human skin in vivo. Microcirculation 2004; 11: 249-259.
- Sikiric P, Seiwerth S, Grabarevic Z, et al. Beneficial effect of a novel pentadecapeptide BPC 157 on gastric lesions induced by restraint stress, ethanol, indomethacin, and capsaicin neurotoxicity. Dig Dis Sci 1996; 41: 1604-614.
- Wood JD. The first Nobel prize for integrated systems physiology: Ivan Petrovich Pavlov, 1904. Physiology (Bethesda) 2004; 19: 326-330.
- Veljaca M, Lesch CA, Sanchez B, Low J, Guglietta A. Protection of BPC-15 on TNBS-induced colitis in rats: possible mechanisms of action. Gastroenterology 1995; 108: 936.
- Veljaca M, Lesch CA, Pllana R, Sanchez B, Chan K, Guglietta A. BPC-15 reduces trinitrobenzene sulfonic acid-inducedcolonic damage in rats. J Pharmacol Exp Ther 1994; 272: 417-422.
- Orsolic N, Seiwerth S, Sikiric P. BPC 157 enhances function of immunological effector cells in mice. J Physiol Pharmacol 2009; 60 (Suppl. 2): 69.
- Szabo S, Deng X, Khomenko T, Chen L, et al. New molecular mechanisms of duodenal ulceration. Ann NY Acad Sci 2007; 1113: 238-255.
- Wallace JL, Cirino G, De Nucci G, McKnight W, MacNaughton WK. Endothelin has potent ulcerogenic and vasoconstrictor actions in the stomach. Am J Physiol 1989; 256: G661-G666.
- Celinski K, Dworzanski T, Fornal R, et al. Comparison of anti-inflammatory properties of peroxisome proliferator-activated receptor gamma agonists rosiglitazone and troglitazone in prophylactic treatment of experimental colitis. J Physiol Pharmacol 2013; 64: 587-595.
- Fagundes DS, Grasa L, Gonzalo S, et al. Mechanism of action of Trolox on duodenal contractility. J Physiol Pharmacol 2013; 64: 705-710.
A c c e p t e d : May 8, 2015